Our Regulation A+ Offering is now officially closed. Please continue to follow our progress at www.insitubiologics.com, on social media, or via newsletter. Thank you.
You are here
Updates
Insitu Biologics continues to make rapid progress towards our mission of providing patients and physicians with an opioid-free solution for post-operative pain. We have added exceptional talent to our team, are nearing manufacturability of clinical-grade product, are in communication with the FDA regarding our clinical strategy moving forward, and have engaged in fundraising on several fronts. Thank you to everyone who has invested in this disruptive technology to curb the opioid crisis and provide a better safer solution for patients in post-operative pain.
For any final investments in our company via the open Reg A+ (click here to invest). Please take some time to read our company update by clicking the preview image below:
(click the preview image above to read)
Click "INVEST NOW" on our offering page, provide your best email address and the platform will walk you through the rest of the process.
If you've already completed your investment, we thank you very much for your contribution, belief in our mission, and trust in our Company! Keep an eye out for our future emails regarding InSitu Biologics' next steps and progress.
As of October 19th, our Stock Offering is once again open and accepting investments. Our plan is to close our Regulation A+ Offering for the last time on Nov 30, 2019.
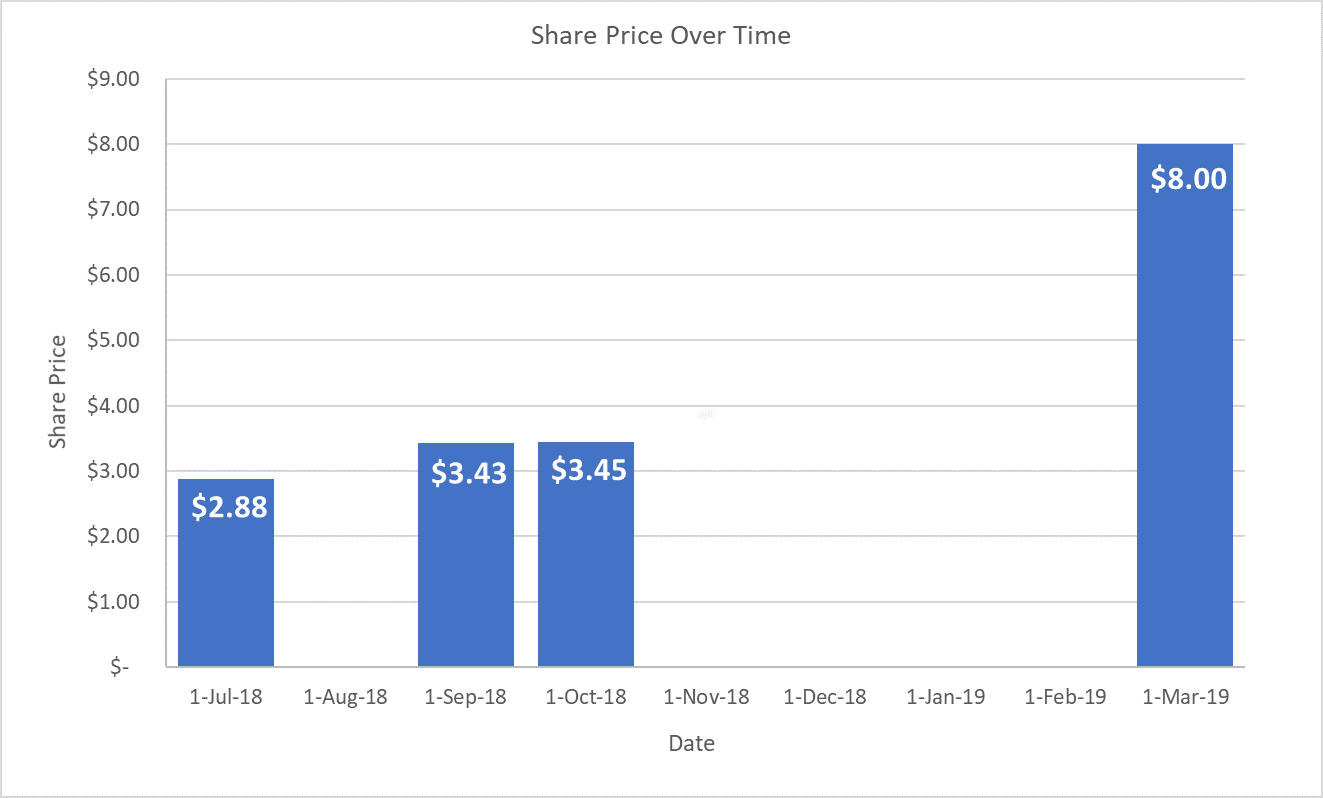
The share price remains at $8.20 and prospective investors may begin the investment process via the green INVEST button on the Offering homepage.
For additional information on the state of the Company and plans for the future, please read the following letter from InSitu Biologics' President and CEO, Kevin Bassett:
As of October 15, our Stock Offering is temporarily paused. During this pause, we will be taking reservations and encourage prospective investors to reserve shares via the green Make Reservation button on the offering home page.
Both prospective and current investors will be updated via The Offering page, insitubiologics.com as well as by email message for those signed up to receive our email correspondence, once The Offering is once again accepting investments.
InSitu Biologics Announces New Leadership
InSitu Biologics today announced that its Board of Directors has appointed Kevin Bassett as President and Chief Executive Officer and member of the Board of Directors effective September 1, 2019 as James Segermark steps down as Chief Executive Officer.
InSitu Biologics has also announces that Dr. Robert Wilson has joined the Board of Directors, effective August 28, 2019.
Kevin Bassett, M.B.A. – President and CEO
Kevin Bassett joined InSitu Biologics in September 2019 as President and CEO. Prior to joining InSitu, Mr. Bassett was General Manager of HLT Medical, a clinical-stage company developing a next-generation heart valve in the field of Transcatheter Aortic Valve Replacement (TAVR). Kevin has also served in a variety of senior executive roles in early-stage medical technology companies over the past twenty years.
Robert Wilson, MD – Member of the Board of Directors
Dr. Wilson has dedicated his professional life to developing new methods for diagnosing and treating heart disease, and to training new physicians. He presently directs the University of Minnesota interventional-cardiology fellowship program and University of Minnesota Physicians clinical cardiovascular services.
Current Report Pursuant to Regulation A
The Company has prepared form 1-U in compliance with the U.S. Securities and Exchange Commission. This document is available to view and we encourage all shareholders to review and save immediately.
The Company is poised for tremendous growth in the coming months as we head into clinical trials with the addition of these new leaders, and we thank you for being a part of our journey.
Regards,
James Knapp
Chairman of the Board
InSitu Biologics
[email protected]
Offering Circular
An offering statement regarding this offering has been filed with the SEC. The SEC has qualified that offering statement, which only means that the company may make sales of the securities described by the offering statement. It does not mean that the SEC has approved, passed upon the merits or passed upon the accuracy or completeness of the information in the offering statement. You may obtain a copy of the offering circular that is part of that offering statement from here.
https://www.manhattanstreetcapital.com/sites/default/files/Insitu_Offering_Circular_10-19-19.pdf
You should read the offering circular before making any investment.
The offering materials may contain forward-looking statements and information relating to, among other things, Insitu Biologics, its business plan and strategy, and its industry. These forward-looking statements are based on the beliefs of, assumptions made by, and information currently available to the company’s management. When used in the offering materials, the words “estimate,” “project,” “believe,” “anticipate,” “intend,” “expect” and similar expressions are intended to identify and constitute forward-looking statements. These statements reflect management’s current views with respect to future events and are subject to risks and uncertainties that could cause the company’s actual results to differ materially from those contained in the forward-looking statements. Investors are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date on which they are made. The company does not undertake any obligation to revise or update these forward-looking statements to reflect events or circumstances after such date or to reflect the occurrence of unanticipated events.
Securities are offered through Sageworks Capital, LLC., a registered broker-dealer and member FINRA/SIPC. Neither InSitu Biologics, Inc. nor Sageworks Capital, LLC provides any investment advice or make any investment recommendations to any persons, ever, and no communication through this website or in any other medium should be construed as such.
InSitu Biologics, Inc. securities are not publicly traded and are intended for investors who do not need a liquid investment. This investment is NOT a bank deposit (and thus NOT insured by the FDIC or by any other federal governmental agency), are NOT guaranteed by Sageworks Capital, LLC or InSitu Biologics, Inc., and MAY lose value. Investors must be able to afford the loss of their entire investment.
Any financial projections or returns shown on the website are illustrative examples only, and there can be no assurance that any valuations provided are accurate or in agreement with market or industry valuations. Investors should conduct their own due diligence, not rely on the financial assumptions or estimates displayed on this website, and are encouraged to consult with a financial advisor, attorney, accountant, and any other professional that can help you to understand and assess the risks associated with any investment opportunity. Please refer to the offering circular for full details and disclosures, including a subscription agreement should you choose to invest.
Insitu Biologics, Inc. is required to pay an advisory retainer fee of $6,000, plus the same value of warrants at the respective issue price. In addition, they will pay a technology admin and service fee of $25 per investor, plus the same value of warrants at the respective issue price. As of 11/30/2018 Insitu Biologics has paid $24,000 in fees.

































Comments